A block has a mass of 20 g and a volume of 5 cm³. What is its density?
Density, Mass and Volume
Density, mass and volume are connected through a formula that allows you to calculate one when the others are known. Density is a type of compound measure, and accurate answers often require careful use of units and conversions.
Overview
Density, mass and volume are connected by one important formula.
If you know any two values, you can work out the third.
What you should understand after this topic
- Understand what density, mass and volume represent
- Use the density formula correctly
- Rearrange the formula to find missing values
- Use correct units in calculations
- Avoid common mistakes in exam questions
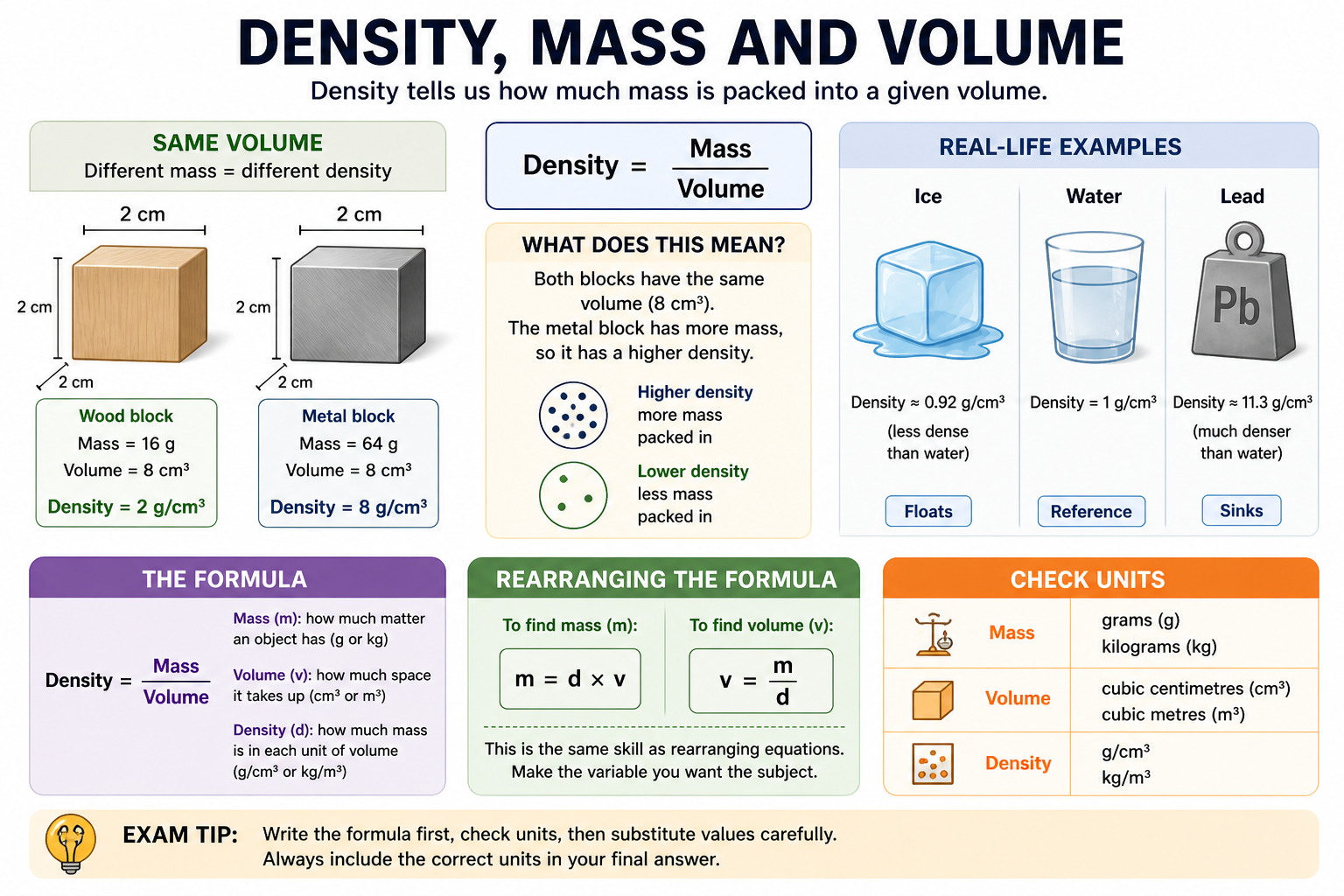
Density
Mass
Volume
Key Definitions
Density
How much mass there is in each unit of volume.
Mass
How much matter an object contains.
Volume
The amount of space an object takes up.
\( \text{cm}^3 \)
Cubic centimetres, a common unit for volume.
\( \text{m}^3 \)
Cubic metres, used for larger volumes.
\( \text{g/cm}^3 \)
A common unit for density.
Key Rules
Main formula
\( \text{Density} = \frac{\text{Mass}}{\text{Volume}} \)
Find mass
\( \text{Mass} = \text{Density} \times \text{Volume} \)
Find volume
\( \text{Volume} = \frac{\text{Mass}}{\text{Density}} \)
Use matching units
Always make sure the units fit together.
Common Units
Mass
g or kg
Volume
\( \text{cm}^3 \) or \( \text{m}^3 \)
Density
\( \text{g/cm}^3 \) or \( \text{kg/m}^3 \)
1 kg
\(1000\) g
1 m³
\(1{,}000{,}000\) cm³
Check first
Do not mix g with \( \text{kg/m}^3 \) without converting.
How to Solve
The main idea
Density describes how much mass is packed into a given volume. It is a type of compound measure.A higher density means more mass is packed into the same space.
How to choose the correct formula
Find density
Divide mass by volume.
Find mass
Multiply density by volume.
Find volume
Divide mass by density.
Check units
Make sure all units match before calculating.
Rearranging the formula
You may need to rearrange the formula depending on what you are finding.
Units matter
Always check that the units are consistent before calculating.
Step-by-step exam method
- Identify what the question is asking for.
- Write the correct formula.
- Check and convert units if needed.
- Substitute the values carefully.
- Calculate the answer.
- Write the final answer with correct units.
Example Questions
Edexcel
Exam-style questions inspired by Edexcel GCSE Mathematics, focusing on calculating density, mass, and volume using standard units.
A block has a mass of 240 g and a volume of 30 cm³. Calculate its density.
A metal cube has a density of 8 g/cm³ and a volume of 15 cm³. Find its mass.
An object has a mass of 540 g and a density of 6 g/cm³. Calculate its volume.
A container holds 2 m³ of a liquid with a density of 750 kg/m³. Find the mass of the liquid.
A solid has a mass of 1.2 kg and a volume of 0.0005 m³. Calculate its density.
AQA
Exam-style questions based on the AQA GCSE Mathematics specification, emphasising unit conversions and multi-step calculations.
A rectangular block has a mass of 3.6 kg and a volume of 0.002 m³. Calculate its density in kg/m³.
A material has a density of 2.5 g/cm³. Find the mass of 120 cm³ of the material.
A substance has a density of 1.2 g/cm³. Calculate the volume of 600 g of the substance.
Convert 2500 kg/m³ to g/cm³.
Explain why units must be consistent when calculating density.
OCR
Exam-style questions aligned with OCR GCSE Mathematics, focusing on reasoning, real-life applications, and compound measures.
A stone has a mass of 90 g and a volume of 36 cm³. Calculate its density.
A tank contains 0.75 m³ of oil with a density of 800 kg/m³. Find the mass of the oil.
A block of wood has a density of 600 kg/m³ and a volume of 0.02 m³. Calculate its mass.
An object has a mass of 500 g and a density of 2 g/cm³. Determine its volume.
Describe the steps required to calculate density when given mass and volume.
Exam Checklist
Step 1
Decide whether you need density, mass or volume.
Step 2
Choose the correct formula or rearrangement.
Step 3
Check whether the units are consistent.
Step 4
Write the final answer with correct units.
Most common exam mistakes
Wrong rearrangement
Using division instead of multiplication, or the other way around.
Wrong units
Writing g when the answer should be \( \text{g/cm}^3 \).
Conversion mistake
Not converting kg to g, or \( \text{m}^3 \) to \( \text{cm}^3 \) when needed.
No final check
Always check if the answer makes sense for the question.
Common Mistakes
These are common mistakes students make when working with density, mass and volume in GCSE Maths.
Using the wrong formula or rearrangement
A student uses the density formula incorrectly when trying to find mass or volume.
Start from the correct formula \(\text{density} = \frac{\text{mass}}{\text{volume}}\) and rearrange carefully depending on what you need to find.
Forgetting units in the answer
A student gives a numerical answer without any units.
Always include the correct units, such as g/cm³ or kg/m³. Marks are often awarded for units.
Mixing grams and kilograms
A student uses grams and kilograms in the same calculation.
Convert all masses to the same unit before calculating. For example, 1 kg = 1000 g.
Mixing volume units
A student uses \(\text{cm}^3\) and \(\text{m}^3\) together without converting.
Volume units must match. Remember that \(1\text{ m}^3 = 1{,}000{,}000\text{ cm}^3\), so convert carefully before calculating.
Multiplying instead of dividing
A student multiplies when calculating density instead of dividing.
Check the formula direction: density = mass ÷ volume. Always confirm whether to multiply or divide based on the rearranged formula.
Try It Yourself
Practise solving problems involving density, mass and volume.
Foundation Practice
Use the density formula to calculate mass, volume and density.
A material has a mass of 60 g and a volume of 10 cm³. Find its density.
A liquid has a density of 2 g/cm³ and a volume of 10 cm³. What is its mass?
A substance has density 5 g/cm³ and volume 4 cm³. Find the mass.
An object has mass 30 g and density 3 g/cm³. What is its volume?
A material has mass 45 g and density 9 g/cm³. Find the volume.
Which formula is correct?
A block has mass 80 g and volume 20 cm³. Find the density.
A student calculates density by doing volume ÷ mass. What is wrong?
A liquid has density 3 g/cm³ and volume 6 cm³. Find the mass.
Higher Practice
Solve density problems involving unit conversions and multi-step reasoning.
A block has mass 2 kg and volume 0.5 m³. What is its density in kg/m³?
A material has density 8 g/cm³ and mass 64 g. Find the volume.
A liquid has density 0.8 g/cm³. What is the mass of 50 cm³?
A block has mass 120 g and volume 30 cm³. Find its density.
A material has density 5 g/cm³. What volume has mass 200 g?
A block has mass 3 kg and density 6 kg/m³. Find the volume.
A student says density = volume ÷ mass. Why is this wrong?
A material has density 2.5 g/cm³ and volume 40 cm³. Find the mass.
A cube has volume 8 cm³ and mass 32 g. What is its density?
A substance has density 0.5 g/cm³ and mass 25 g. Find the volume.
Games
Practise this topic with interactive games.